
Pluvicto is a PSMA-Targeted Radiopharmaceutical Therapy for Prostate Cancer
Pluvicto is a form of theranostics, which is a treatment approach that combines diagnostics and therapy, using radioactive drugs (radiopharmaceuticals) to deliver targeted treatment to cancer cells.
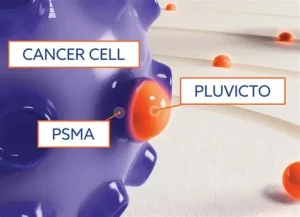
What is Radiopharmaceutical Therapy?
Radiopharmaceutical therapy is a targeted cancer treatment that uses radioactive drugs to destroy cancer cells. The drug is delivered intravenously, or orally, where it travels through the blood stream to find and bind to specific tumors, killing the cancer cells.
What is PSMA?
PSMA stands for prostate-specific membrane antigen, and is a protein found in high amounts on the surface of most prostate cancer cells. A PSMA PET/CT is a highly sensitive imaging test that reveals the exact location of prostate cancer by using a radioactive tracer that binds to the prostate-specific membrane antigen, found on the surface of prostate cancer cells. The PSMA PET/CT scan can detect prostate cancer throughout the body, particularly for identifying whether the prostate cancer has spread (metastasized) or recurred after treatment.
What is mCRPC diagnosis?
mCRPC is metastatic castration-resistant prostate cancer. This diagnosis is an advanced, incurable state of prostate cancer that has spread to other parts of the body (metastatic) and continues to grow, despite hormone therapy lowering testosterone to very low levels (castration-resistant). Someone with this diagnosis may be a candidate for Pluvicto.
What is Pluvicto?
Pluvicto (Luticium-177 vipivotide tetraxetan) is an FDA-approved, targeted radiogland therapy for PSMA-positive metastatic castration-resistant prostate cancer (mCRPC). Pluvicto combines a targeting molecule with radioactive material to deliver radiation directly to cancer cells intravenously, binding to the PSMA protein found on the surface of the cancer cells, and killing the cancer cells while minimizing damage to surrounding healthy tissue. The Pluvicto treatment reduces tumor size and slows disease progression, after other treatments have failed.
Who is a candidate for Pluvicto?
Pluvicto is prescribed to treat adults with prostate-specific membrane antigen (PSMA) – prostate metastatic castration-resistant prostate cancer (PSMA-positive mCRPC) who have already been treated with:
- Hormone therapy and are considered appropriate to delay chemotherapy, or
- Hormone therapy and chemotherapy
What to Expect as a Pluvicto patient?
Patients who qualify for Pluvicto are scheduled for a consultation, where all details are reviewed and discussed about the patient’s diagnosis and the effectiveness and side effects of the medication. In addition, patients and caregivers are made aware of all steps to prepare for pre-treatment, during treatment and post treatment. Pluvicto is administered via intravenous (IV) infusion with one dose every 6 weeks for up to 6 doses, with lab work required prior to each dose. Appointment times are scheduled for one hour, however, often patients do not require that much time.